Rules: As usual this is for the UK version of the BMJ. The classified advertisement section is excluded, as are pages advertising the BMA or products of the BMJ/BMA/BNF.
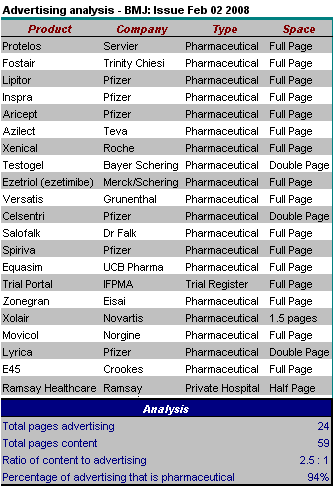
Comment: There were 24.5 pages of advertising (1 page of advertising for each 2.5 pages of content). The proportion of advertising devoted to pharmaceuticals fell to an all time low of 94%. The other 4% was:
- a half page advertisement for Ramsay Healthcare (for more about Ramsay and their cozy links with the Labour Government see here)
- a one page advertisement for a register of clinical trials.
There were a few other notable items this issue:
- A letter from a reader (Jeremy Stone) complaining about discordance between a double page advertisement for testosterone supplements in the previous issue, and (on the very next page) a double blind RCT showing that testosterone supplements have no effect on functional mobility, muscle strength, cognitive function, bone mineral density or quality of life. I'll look out for proper referencing in the next appearance of this advertisement. They might even want to reference our paper showing that testosterone supplementation does nothing useful to anything (except plasma testosterone).
- There was a dismal letter signed by around 20 statisticians asserting that the EU regulatory agencies should employ more card carrying statisticians to "explore methods and data in real depth". The plea for employment and the special God-given powers of statisticians would carry more credibility if there were evidence that any of the signatories were regular commentators on instances of blatant fraud in clinical trials (no statistical expertise required) or on the fact that neither regulators nor scientific authors have genuine raw data to "explore" in any case. None (as far as I am aware) have commented on Ezetimibe. Statisticians would also do well to realize that only a few aspects of data integrity are within the special remit of complex statistics. All scientists require raw data.
- There was a detailed Obituary of Moses Judah Folkman (the "father of angiogenesis") by Jeanne Lenzer. In contrast to other Folkman obituaries it did at least mention that he had been forced to "apologise" over scientific fraud (false descriptions of methodology and falsified findings in an NEJM publication).
Click here for collated BMJ Advertising analyses.
Earlier|Later|Main Page
1 comment:
Economic Left/Right: -5.25
Social Libertarian/Authoritarian: -5.64
There were too many questions however where "agree" or "disagree" are forced on you and where neither reflects an accurate view.
It needs at least a choice in the center - even if its a simple as "I don't know" or "It depends on circumstances/conditions".
Interesting to do - but the reliability of the total score has to be questionable given in places there is no choice but to plump for one of two unsatisfactory answers.
Post a Comment